Blog
Database Update!
Our databases page has been updated and a new database has been published! Find below descriptions of the information available in each database: Prevalence: Prevalence and incidence rates for different…
Read MoreGrant Opportunity: 2020 Innovator Awards
The Hydrocephalus Association requests applications for: 2020 INNOVATOR AWARDS Innovator Award Research Focus: Goal: To provide seed funding for high-quality, innovative research that potentially improves hydrocephalus treatments and outcomes or…
Read MoreHydrocephalus Society: Call for Abstracts
Call for Abstracts! We are pleased to announce the Call for Abstracts of the Thirteenth Meeting of the Hydrocephalus Society, which will be held in Gothenburg, Sweden from 11 to 14 September 2020! Don’t miss the opportunity to present your research and outcomes and have substantive…
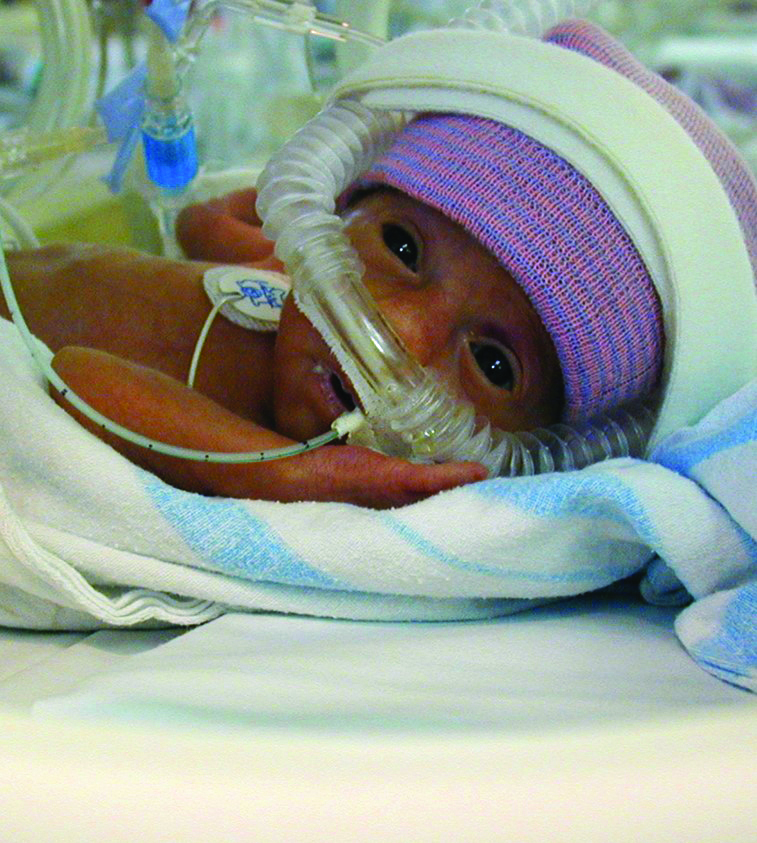
Read MoreNewest Results from ELVIS: Early vs. Late Ventricular Intervention
Newest Results from the Early vs. Late Ventricular Intervention Study (ELVIS) in Preterm Infants with Posthemorrhagic Ventricular Dilation By: Rebecca A. Dorner, MD MHS Neonatology Fellow Johns Hopkins Hospital The…
Read MoreKeystone Symposia Announcement: February 16-19, 2020
KEYSTONE SYMPOSIA Cerebral Fluid Flow and Function: Lymphatics, Glymphatics and the Choroid Plexus Scientific Organizers: Edwin S. Monuki, Maria Lehtinen, and Maiken Nedergaard Eldorado Hotel & Spa,Sante Fe, New Mexico…
Read MoreHydrocephalus Society – Call for Abstracts
Call for Abstracts! The final date for the Abstract Submission for the Eleventh Meeting of the International Society for Hydrocephalus and Cerebrospinal Fluid Disorders, which will be held in…
Read MorePhysician Scientist Fellowship: Doris Duke Charitable Foundation Announcement
The Doris Duke Charitable Foundation Announces a New Award for Subspecialty Fellows 2019 Physician Scientist Fellowships The Doris Duke Charitable Foundation (DDCF) today announced the creation of the Doris…
Read MoreJoin the SRHSB in Spain! Abstracts due February 15th
The 63rd Annual Meeting of the SRHSB La Laguna, Canarias on 26-29 June 2019. Abstract Deadline: February 15th Learn More: https://www.srhsb.com/la-laguna-2019 The abstract submission deadline for the 63rd Annual Meeting of…
Read MoreGrant Opportunity! 2019 DoD CDMRP PRMRP Announcement
Hydrocephalus research eligible for CDMRP PRMRP Funding Congress appropriated $350 million for the FY19 program to solicit proposals in 49 topic areas. This week the Congressionally Directed Medical Research Program…
Read More2018 Discovery Science Award Grantees for PHH Announced
ANNOUNCING… The 2018 Discovery Science Award Recipients for Posthemorrhagic Hydrocephalus! A big thank you to all of the applicants and reviewers of the 2018 Discovery Science Award (DSA) for Posthemorrhagic…
Read More